| International Journal of Clinical Pediatrics, ISSN 1927-1255 print, 1927-1263 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, Int J Clin Pediatr and Elmer Press Inc |
| Journal website https://ijcp.elmerpub.com |
Review
Volume 000, Number 000, May 2026, pages 000-000
Associated Arthropathies in Pediatric Endocrine Disorders: A Systematic Review and Narrative Synthesis
Ashraf T. Solimana, b , Mohammed Qusada, Mohamed Alkalafa, Khaled A. Siddiqa, Fawzia Alyafeia, Nada Alaaraja, Noor Hameda, Shayma Ahmeda
aDepartment of Pediatrics, Division of Endocrinology, Hamad Medical Corporation, Doha, Qatar
bCorresponding Author: Ashraf T. Soliman, Department of Pediatrics, Hamad Medical Corporation, PO Box 3050, Doha, Qatar
Manuscript submitted January 26, 2026, accepted March 20, 2026, published online May 11, 2026
Short title: Pediatric Endocrine Arthropathies
doi: https://doi.org/10.14740/ijcp1044
| Abstract | ▴Top |
Background: Endocrine disorders in children and adolescents frequently involve the musculoskeletal system, producing characteristic arthropathies that may precede the diagnosis of the underlying hormonal disease. These manifestations are often overlooked despite their diagnostic value and potential reversibility with timely treatment. Improved recognition of endocrine-related arthropathy is essential to optimize diagnosis, management, and long-term functional outcomes. The objectives of the study were to summarize the spectrum and prevalence of arthropathy in pediatric endocrine disorders, evaluate their diagnostic and pathophysiological significance, and discuss management strategies and prognosis.
Methods: A systematic search of PubMed and Scopus was conducted from database inception through January 1, 2026 using combinations of endocrine terms (type 1 diabetes, thyroid disorders, growth hormone (GH) disorders, Cushing syndrome, hyperparathyroidism, and pseudohypoparathyroidism) and musculoskeletal terms (arthropathy, limited joint mobility, joint stiffness, slipped capital femoral epiphysis (SCFE), bone pain, and fractures). Titles, abstracts, and full texts were screened independently by two reviewers according to predefined criteria, and the study-selection pathway was summarized in a PRISMA-style flow diagram. Pediatric studies were prioritized; selected mixed-age cohorts were retained only when they included adolescents or informed rare pediatric phenotypes. Methodological quality was assessed using Cochrane risk-of-bias principles adapted for mixed study designs, supplemented by the Newcastle–Ottawa Scale for observational studies. Because of substantial heterogeneity, a narrative synthesis was performed.
Results: Fifteen studies were included. Diabetic arthropathy, usually presenting as limited joint mobility (cheiroarthropathy), was the most common finding, affecting up to 30% of children in earlier cohorts but generally < 10% in contemporary populations with improved glycemic control; it correlated with disease duration and microvascular complications. Thyroid disorders showed variable joint involvement: hypothyroidism was associated with arthralgia, non-inflammatory effusions, epiphyseal dysgenesis, and atypical SCFE, whereas Graves disease rarely caused thyroid acropachy or antithyroid drug–related arthritis. GH excess was associated with acromegalic arthropathy involving the hands, spine, and large joints, although prevalence estimates of 70–75% derive mainly from mixed-age cohorts that included adolescents rather than pediatric-only series. In contrast, GH deficiency and hypothyroidism were overrepresented among children with atypical or early-onset SCFE. Pediatric Cushing syndrome was characterized mainly by bone fragility, vertebral fractures, and reduced mobility rather than inflammatory arthritis. Primary hyperparathyroidism caused bone and joint pain with fractures, while pseudohypoparathyroidism was associated with growth-plate abnormalities and occasional SCFE. Across disorders, early endocrine treatment usually stabilized or improved musculoskeletal manifestations.
Conclusions: Arthropathy in pediatric endocrine disorders encompasses a broad, disorder-specific spectrum with important diagnostic and prognostic implications. Early recognition and treatment of the underlying endocrinopathy can prevent or reverse many joint complications, whereas delayed diagnosis may lead to permanent musculoskeletal disability. Multidisciplinary management is essential to optimize long-term outcomes.
Keywords: Pediatric endocrinology; Arthropathy; Limited joint mobility; Slipped capital femoral epiphysis; Growth hormone disorders; Systematic review
| Introduction | ▴Top |
Musculoskeletal complaints, including arthralgia, joint stiffness, reduced range of motion, and structural joint abnormalities, are increasingly recognized as relevant comorbidities in children and adolescents with endocrine disorders [1, 2]. Although traditionally considered secondary or minor manifestations, arthropathy in pediatric endocrinology has important clinical implications, as it may affect mobility, physical activity, growth-related function, and quality of life during critical developmental periods [1–3]. In some cases, joint manifestations precede the diagnosis of the underlying endocrine disorder, making them valuable early clinical indicators [2, 4].
The reported prevalence of endocrine-related arthropathy varies widely across conditions and populations. Among pediatric endocrine disorders, type 1 diabetes mellitus represents the most extensively studied model. Limited joint mobility (LJM) of the hands was historically reported in up to 30–40% of children and adolescents with diabetes [5–7]. More recent cohorts, however, demonstrate substantially lower prevalence—often below 10%—likely reflecting improvements in long-term glycemic control, earlier diagnosis, and advances in diabetes management [8, 9]. These observations highlight the dynamic nature of endocrine arthropathy and its sensitivity to disease duration and metabolic control.
Beyond diabetes, musculoskeletal involvement has been described across a broad spectrum of endocrine disorders in children, including conditions affecting thyroid function, growth hormone (GH) secretion, and mineral metabolism. While the specific patterns and mechanisms differ, shared pathways—such as altered collagen metabolism, glycation of connective tissue, hormonal effects on cartilage and bone turnover, and impaired microvascular supply—appear to underlie joint vulnerability in endocrine disease [1, 2, 10]. Importantly, endocrine disorders are overrepresented among children presenting with atypical orthopedic conditions, including slipped capital femoral epiphysis (SCFE) occurring outside the typical age or weight range, reinforcing the link between hormonal imbalance and joint pathology [10].
The clinical expression of endocrine-related arthropathy in children differs fundamentally from that in adults due to ongoing growth, open physes, and rapid changes in body composition during childhood and adolescence [1, 2]. These developmental factors influence not only susceptibility to joint involvement but also the potential for reversibility following timely treatment. While some joint manifestations improve with optimal endocrine control, others may persist or progress, particularly when diagnosis is delayed or exposure to hormonal imbalance is prolonged [3, 9].
Despite its clinical relevance, arthropathy remains under-recognized in routine pediatric endocrine practice. Clinical evaluations often prioritize auxological parameters, pubertal staging, and biochemical targets, with less systematic attention to joint examination and functional assessment [1, 3]. This may result in delayed recognition, missed opportunities for early intervention, and underestimation of disease burden, especially in children with subtle or painless joint involvement.
Accordingly, a focused review of arthropathy in endocrine disorders among children and adolescents is warranted. By summarizing prevalence patterns and clinical presentations using validated evidence, this review aims to improve awareness and early recognition of musculoskeletal involvement in pediatric endocrinology. Condition-specific mechanisms, diagnostic considerations, and management strategies are addressed in detail in subsequent sections, with the goal of optimizing multidisciplinary care and improving long-term musculoskeletal outcomes.
Objectives
The objectives were: 1) to summarize the prevalence and clinical patterns of arthropathy associated with major endocrine disorders in children and adolescents; 2) to identify endocrine conditions in which joint manifestations serve as early diagnostic or prognostic indicators; and 3) to synthesize current evidence on outcomes following endocrine-specific management, with emphasis on reversibility and long-term musculoskeletal impact.
| Methods | ▴Top |
Literature search and study selection
A structured literature search was performed in PubMed (MEDLINE) and Scopus from database inception through January 1, 2026 to identify reports of arthropathy or joint-related musculoskeletal manifestations in children and adolescents with endocrine disorders. The core search strategy combined endocrine terms (“type 1 diabetes”, “hypothyroidism”, “hyperthyroidism”, “Graves disease”, “growth hormone excess”, “pituitary gigantism”, “acromegaly”, “growth hormone deficiency”, “Cushing syndrome”, “hyperparathyroidism”, and “pseudohypoparathyroidism”) with musculoskeletal terms (“arthropathy”, “arthritis”, “arthralgia”, “limited joint mobility”, “cheiroarthropathy”, “joint stiffness”, “slipped capital femoral epiphysis”, “bone pain”, “fracture”, and “musculoskeletal”). Boolean operators AND/OR were used, and the reference lists of relevant review articles were hand-searched for additional eligible reports.
Searches were restricted to human studies and English-language publications. Pediatric studies (0–18 years) were prioritized. Because several endocrine arthropathies are rare in childhood, selected mixed-age cohorts were also retained when they included adolescents or when pediatric-specific evidence was limited but the study directly informed mechanisms, phenotype, or management relevant to pediatric disease.
Eligible studies included original research articles of any design (randomized trials, cohort studies, case–control studies, cross-sectional studies, case series, and single-case reports) that described joint or skeletal manifestations in the context of a clearly defined endocrine disorder in pediatric patients. Review articles, editorials, and textbook chapters were excluded as primary evidence but were used for background context. Studies focused on primary rheumatologic disorders or non-endocrine metabolic bone diseases were excluded.
After duplicate removal, titles and abstracts were independently screened by two reviewers using predefined inclusion and exclusion criteria. Potentially eligible full texts were assessed independently, and disagreements were resolved by consensus or by consultation with a third reviewer when required. The study-selection pathway is summarized in a PRISMA-style flow diagram (Fig. 1).
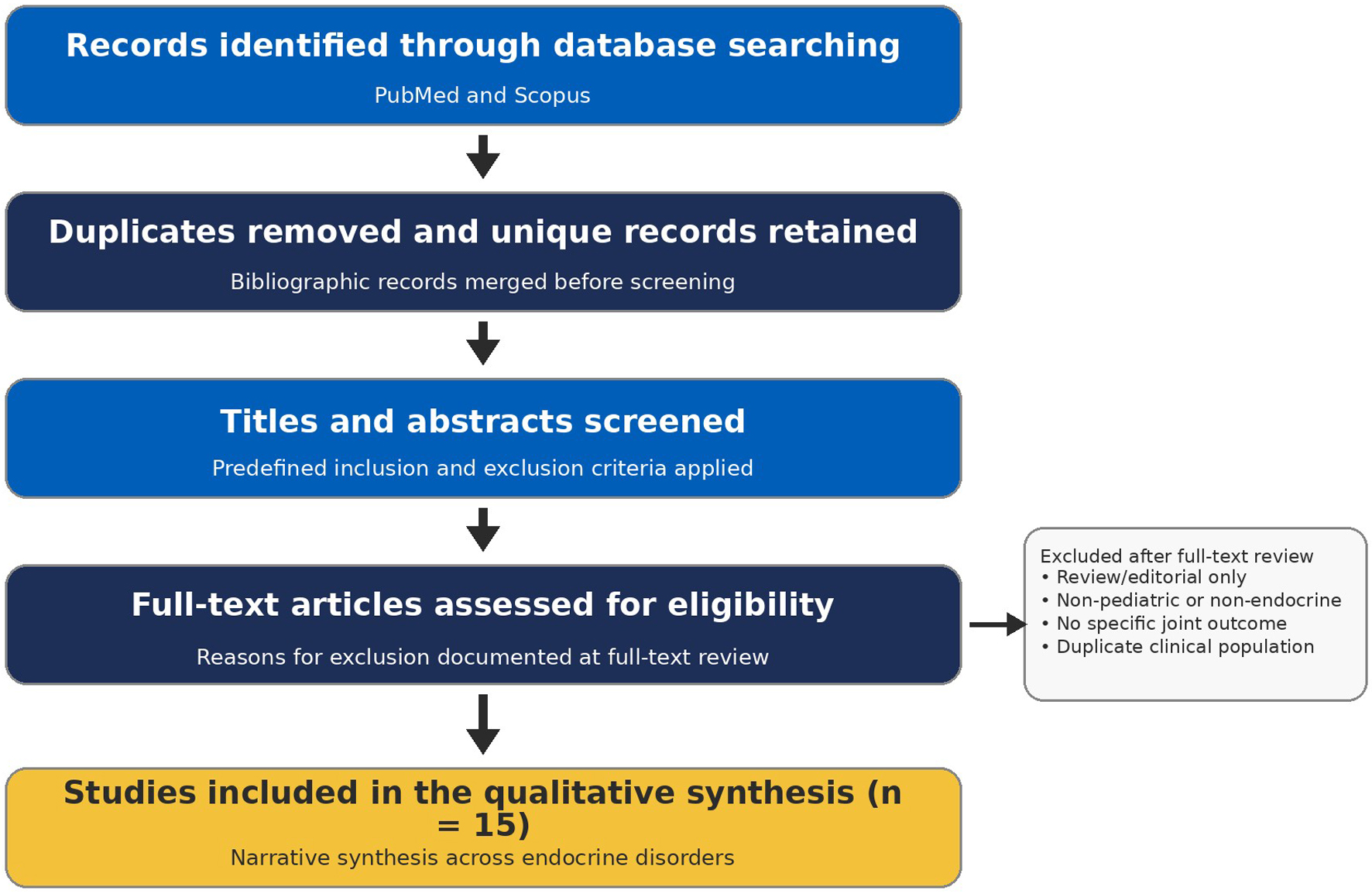 Click for large image | Figure 1. PRISMA-style study-selection pathway for the systematic review. This PRISMA-style figure summarizes database searching, duplicate removal, screening, full-text eligibility assessment, and final inclusion of 15 studies in the qualitative synthesis. |
Data extraction
From each included study, the following data were extracted using a standardized form: study design, sample size, age range, endocrine diagnosis, type of arthropathy or joint involvement, reported prevalence or frequency, diagnostic methods, and clinical outcomes following endocrine treatment.
For observational studies, reported associations between arthropathy and clinical variables (e.g., disease duration, metabolic control) were recorded. For interventional or longitudinal studies, changes in joint manifestations after endocrine therapy were documented. Case reports and case series were included to capture rare but clinically relevant presentations. Extracted data were organized by endocrine disorder and summarized in thematic tables.
Quality assessment
Methodological quality and risk of bias were assessed using an adapted Cochrane risk-of-bias framework, appropriate for mixed study designs.
For cohort and case–control studies, the Newcastle–Ottawa Scale was used as a supplementary guide. Case series and case reports were recognized as having inherent high risk of bias but were retained for descriptive purposes due to the rarity of several endocrine arthropathies. Two reviewers independently rated each study as having low, moderate, or high risk of bias, with disagreements resolved by consensus. A summary quality assessment table is provided.
Given the heterogeneity of endocrine disorders, study designs, and outcome definitions, a narrative synthesis was undertaken. Results were grouped by endocrine condition and type of arthropathy. Prevalence data are presented as ranges where applicable. Descriptive statistics reported in the original studies (percentages, means, confidence intervals) are cited directly.
Formal meta-analysis was not performed due to methodological heterogeneity and limited availability of comparable quantitative data. No additional statistical analyses were conducted beyond those reported in the original studies.
Reporting standards
This systematic review was conducted and reported in accordance with PRISMA 2020 principles. Because the included evidence was clinically heterogeneous and frequently descriptive, the results are presented as a narrative synthesis. As this review was based exclusively on published literature, institutional review board approval and informed consent were not required.
| Results | ▴Top |
The results synthesize evidence from observational cohorts, registry analyses, and selected case-based reports to characterize the prevalence, clinical patterns, and risk profiles of arthropathy across pediatric endocrine disorders. To reduce overlap, Table 1 [4, 9, 11–22] focuses on study characteristics and the principal clinical message of each included study, whereas Table 2 [4, 11–14, 16–24] summarizes disorder-specific manifestations and reported prevalence. Findings are organized by disorder and mechanism, with emphasis on population-level trends when available and on distinctive clinical phenotypes that inform diagnosis and management.
 Click to view | Table 1. Study Characteristics and Key Clinical Messages of Arthropathy in Pediatric Endocrine Disorders |
 Click to view | Table 2. Disorder-Specific Clinical Manifestations and Reported Prevalence of Arthropathy in Pediatric Endocrine Disorders |
Table 1 summarizes the foundational clinical evidence linking endocrine disorders to pediatric arthropathy, highlighting diabetes as the most extensively studied model and illustrating how improvements in disease management have altered musculoskeletal risk over time. It establishes the heterogeneity of joint involvement across endocrine conditions and underscores the influence of disease duration and metabolic control on arthropathy prevalence.
Table 2 provides a comparative overview of musculoskeletal involvement across major pediatric endocrine disorders, demonstrating wide variability in prevalence and phenotype. The data emphasize that while some conditions (e.g., type 1 diabetes, GH excess) show relatively frequent joint involvement, others are characterized by rare but diagnostically important manifestations, reinforcing the need for disorder-specific clinical vigilance.
Table 3 [5, 6, 9, 11, 25, 26] highlights the temporal decline in LJM prevalence in pediatric diabetes cohorts, paralleling advances in glycemic management. It reinforces the strong association between LJM, disease duration, and microvascular complications, supporting its role as a marker of cumulative metabolic burden rather than an isolated musculoskeletal disorder.
 Click to view | Table 3. Prevalence and Features of Diabetic Arthropathy (Limited Joint Mobility) in Children With Type 1 Diabetes |
Table 4 [4, 14, 20, 23, 27–29] clearly shows that thyroid-related arthropathy in children is usually non-inflammatory and often reflects growth-plate and skeletal maturation effects rather than primary synovitis. Hypothyroidism is linked to arthralgia, viscous effusions, epiphyseal dysgenesis, and atypical SCFE, emphasizing the diagnostic value of thyroid testing in unexplained effusions or SCFE outside the usual age/weight range [4, 14, 27]. In contrast, musculoskeletal problems in Graves disease are rare and typically iatrogenic or periosteal—most often thionamide-related arthritis or very rare thyroid acropachy—so recognizing these patterns helps avoid mislabeling as rheumatologic disease and supports prompt medication adjustment when appropriate [20, 23, 28].
 Click to view | Table 4. Thyroid-Related Arthropathy and Musculoskeletal Manifestations in Children and Adolescents |
Table 5 [30–36] highlights two distinct GH-related musculoskeletal patterns: degenerative arthropathy in GH excess and growth-plate vulnerability (SCFE) in GH deficiency or GH therapy. Pediatric-onset GH excess is associated with periarticular overgrowth and osteoarthritis (OA)-like joint changes that may persist despite biochemical control, whereas GH deficiency and GH treatment primarily increase the risk of SCFE, particularly in children with underlying endocrinopathies, supporting the need for early recognition and orthopedic vigilance.
 Click to view | Table 5. Growth Hormone (GH) Excess– and Deficiency–Related Arthropathy in Children and Adolescents: Key Clinical Patterns and Representative Evidence |
Table 6 [1–36] provides a structured appraisal of methodological quality across the evidence base, demonstrating that most data derive from moderate-quality observational studies, with fewer low-bias population cohorts and necessary reliance on high-bias case reports for rare phenotypes. This assessment contextualizes prevalence estimates and supports cautious interpretation while highlighting areas where prospective pediatric data are most needed.
 Click to view | Table 6. Overall Risk of Bias Across Studies Included in the Review |
Overall, the evidence base for pediatric endocrine arthropathy is dominated by observational studies (moderate risk) with stronger epidemiologic inference available for SCFE in endocrinopathy from large population datasets (low-to-moderate risk). Rare thyroid and parathyroid phenotypes rely largely on case-based evidence (high risk), which is informative clinically but should not be used to generate population prevalence estimates (Fig. 2).
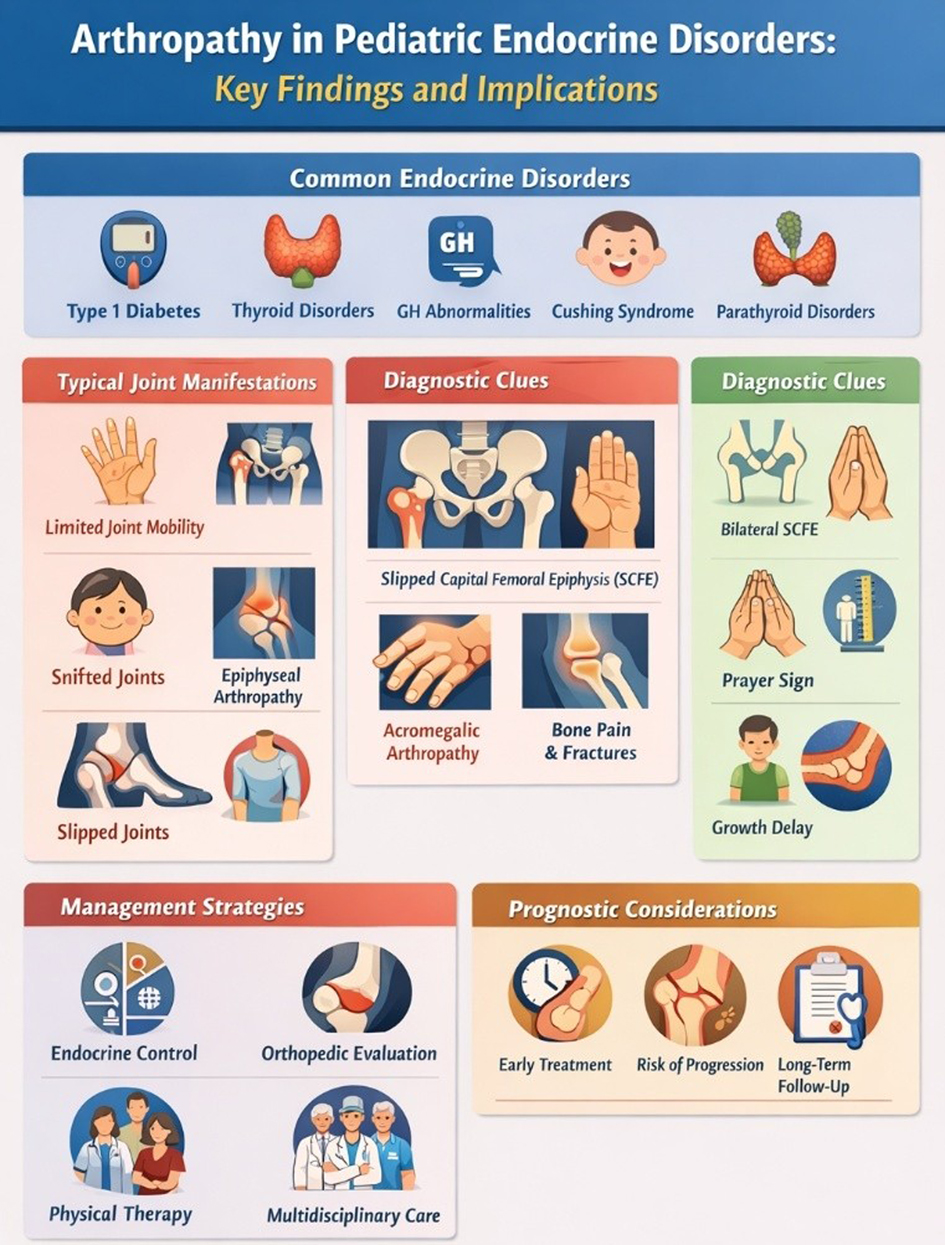 Click for large image | Figure 2. Arthropathy in pediatric endocrine disorders: clinical patterns, diagnostic clues, and management pathways. Infographic summarizing disorder-specific joint manifestations, diagnostic red flags, and management considerations in pediatric endocrine arthropathy. SCFE: slipped capital femoral epiphysis. |
| Discussion | ▴Top |
This review synthesizes available evidence on arthropathy in pediatric endocrine disorders, highlighting that joint and peri-articular involvement represents a clinically relevant but frequently underrecognized component of endocrine morbidity in children and adolescents. Across disorders, arthropathy manifests along a spectrum ranging from subclinical limitation of joint mobility to overt structural joint and growth-plate pathology. The findings from Tables 1–5 collectively address the review objectives by defining prevalence patterns, identifying diagnostic signals, and clarifying implications for prognosis and management.
One consistent theme emerging from the data is that arthropathy often reflects cumulative disease exposure rather than acute metabolic status. LJM was most prevalent in earlier cohorts with less intensive glycemic management and declined substantially in more recent cohorts [5–7, 11, 12]. Importantly, LJM correlated with microvascular complications such as retinopathy and microalbuminuria, supporting its role as a clinical surrogate of long-term tissue glycation and microangiopathy rather than an isolated musculoskeletal condition [5, 9, 12]. This observation aligns with the first objective of summarizing prevalence and patterns, while also reinforcing the prognostic value of joint examination in routine endocrine follow-up.
Mechanistically, diabetic arthropathy is thought to result from non-enzymatic glycation of collagen, leading to increased cross-linking, reduced tissue elasticity, and impaired remodeling within peri-articular connective tissue [13]. Accumulation of advanced glycation end products may also compromise microvascular perfusion of tendons and joint capsules, explaining the parallel association between LJM and microvascular complications observed across multiple cohorts [5, 9, 11, 12]. Clinically, the prayer sign is elicited by asking the patient to oppose the palmar surfaces of both hands fully; inability to bring the fingers and palms into complete apposition supports LJM. Although often asymptomatic, this process can lead to progressive stiffness and reduced range of motion, particularly in the hands, underscoring the practical value of bedside screening in endocrine clinics.
In contrast to diabetes, thyroid-related arthropathy is less prevalent but often diagnostically pivotal. Hypothyroidism in children rarely causes inflammatory arthritis; instead, joint symptoms arise indirectly through delayed skeletal maturation, epiphyseal dysgenesis, and altered cartilage turnover [4, 14, 29]. These changes predispose to atypical SCFE, particularly in children presenting outside the usual pubertal age or weight profile [15, 27]. In this context, “atypical SCFE” refers to bilateral disease, presentation in a younger child or in a non-obese adolescent, short stature, or the presence of additional endocrine/systemic features that lower the threshold for endocrine testing. The association between hypothyroidism and SCFE therefore emphasizes that joint manifestations may serve as early diagnostic indicators of underlying endocrine disease.
Graves disease–related joint manifestations are rare and mechanistically distinct. When present, they are more often related to antithyroid drug reactions or rare periosteal syndromes such as thyroid acropachy rather than direct effects of thyroid hormone excess [20, 23, 28]. Recognition of these patterns is essential to avoid misdiagnosis as primary rheumatologic disease and to guide appropriate management, such as drug withdrawal rather than immunosuppression.
Evidence summarized in Table 5 demonstrates that GH excess, whether presenting as gigantism or acromegaly, is associated with a high burden of arthropathy. Joint involvement in GH excess is characterized by peri-articular soft-tissue overgrowth, cartilage hypertrophy, and subsequent degenerative changes, which may progress despite biochemical control [18, 19, 31–34]. Importantly, the highest prevalence estimates derive mainly from mixed-age cohorts that included adolescents rather than pediatric-only series. Imaging studies indicate that while some cartilage abnormalities may partially regress with treatment, established structural changes often persist, leading to chronic pain and functional limitation [19, 33]. These findings underscore that early diagnosis and endocrine treatment are critical, but they also support early orthopedic assessment, individualized physical therapy, and range-of-motion preservation strategies to reduce secondary disability once symptoms emerge.
In children with GH deficiency or those receiving GH therapy, the dominant arthropathic concern is SCFE rather than intrinsic joint disease [15, 17, 30, 35, 36]. Large registry and population-based cohort studies demonstrate that SCFE risk is elevated in children with endocrinopathies, particularly those with multiple pituitary hormone deficiencies or rapid growth acceleration during therapy [30, 35, 36]. Mechanistically, this vulnerability likely reflects disproportionate physeal stress during periods of accelerated linear growth, compounded by underlying skeletal immaturity or hormonal imbalance. These data support proactive orthopedic surveillance and rapid evaluation of hip or knee pain in at-risk patients.
It must be noted that heterogeneity in study design, outcome definitions, and screening intensity substantially influences reported prevalence. Conditions such as diabetic LJM benefit from multiple cohort studies with relatively consistent methodologies, whereas rarer disorders (e.g., pseudohypoparathyroidism, pediatric hyperparathyroidism) rely largely on case reports and small series [16, 21]. As summarized in the quality assessment (Table 6), most evidence falls into the moderate risk-of-bias category, emphasizing the need for cautious interpretation of prevalence figures and avoidance of overgeneralization.
From a diagnostic perspective, the data reinforce that joint findings should prompt consideration of endocrine etiologies, particularly when presentations are atypical for primary rheumatologic disease. Recognizing these musculoskeletal patterns helps distinguish endocrine-related arthropathy from primary inflammatory arthritis and guides targeted hormonal evaluation. Bilateral SCFE, SCFE outside the usual age range, non-inflammatory joint effusions, or progressive stiffness without synovitis should trigger focused endocrine assessment [1, 2, 10, 15]. Incorporating structured musculoskeletal examination into pediatric endocrine clinics may therefore improve early detection and prevent irreversible complications.
Management of endocrine-related arthropathy remains largely etiology-specific. Optimal endocrine control is foundational but not always sufficient to reverse joint pathology, particularly in GH excess and long-standing hypothyroidism [4, 18, 19, 31–34]. Adjunctive strategies, including physical therapy to preserve range of motion, early orthopedic review for symptomatic hip disease or SCFE risk, and longitudinal monitoring for degenerative change, are often required. Recognizing drug-induced arthropathy in Graves disease is equally important because prompt withdrawal or substitution of the offending antithyroid medication may resolve symptoms without unnecessary escalation to immunosuppressive therapy [23, 28].
Prognosis varies widely across disorders and phenotypes. Diabetic LJM often stabilizes or improves with modern care, whereas GH excess–related arthropathy may progress despite remission [19, 33, 34]. SCFE associated with endocrine disorders carries a higher risk of bilaterality and long-term morbidity [17, 30, 35, 36], reinforcing the importance of early recognition and coordinated care. These distinctions highlight the value of disorder-specific counseling for patients and families.
The findings also expose gaps in the literature, including the lack of standardized definitions for pediatric endocrine arthropathy, limited prospective data linking early joint findings to long-term functional outcomes, and sparse pediatric-specific evidence for uncommon disorders. Future studies that integrate imaging, validated functional assessments, and endocrine biomarkers may better define risk trajectories and inform preventive strategies, particularly for mixed-age conditions in which pediatric estimates currently rely on indirect extrapolation.
| Conclusion | ▴Top |
Arthropathy in children and adolescents with endocrine disorders represents a heterogeneous but clinically significant source of morbidity that extends beyond traditional metabolic and growth outcomes. Evidence from observational cohorts and registry studies demonstrates that joint manifestations may serve as early diagnostic clues, markers of cumulative disease burden, or predictors of long-term functional impairment, depending on the underlying endocrinopathy. While some forms of endocrine-related arthropathy improve with timely hormonal correction, others—particularly those associated with GH excess or growth-plate vulnerability—may persist or progress despite biochemical control. Integrating systematic musculoskeletal assessment into pediatric endocrine care is therefore essential to optimize diagnosis, management, and prognosis.
Recommendations
Acknowledgments
The authors thank the clinical and research staff of the Department of Pediatrics, Hamad Medical Corporation, and the institutional librarians for assistance with article retrieval and administrative support.
Financial Disclosure
This research received no external funding and was supported only by routine institutional resources of the Department of Pediatrics, Hamad Medical Corporation, Doha, Qatar.
Conflict of Interest
The authors declare that they have no conflict of interest relevant to this work.
Informed Consent
Not applicable because no individual participant data were collected directly by the authors.
Author Contributions
ATS conceived the review; MQ, MA, and KAS contributed to study selection and data organization; FA and NA contributed to manuscript review and administrative support; NH and SA assisted data extraction and table preparation; ATS and MQ interpreted the evidence; all authors revised the manuscript critically and approved the final version.
Data Availability
The authors declare that data supporting the findings of this study are available within the article.
| References | ▴Top |
- Boswell SB, Patel DB, White EA, Gottsegen CJ, Forrester DM, Masih S, Matcuk GR, Jr. Musculoskeletal manifestations of endocrine disorders. Clin Imaging. 2014;38(4):384-396.
doi pubmed - Jacobs-Kosmin D, DeHoratius RJ. Musculoskeletal manifestations of endocrine disorders. Curr Opin Rheumatol. 2005;17(1):64-69.
doi pubmed - Chew FS. Radiologic manifestations in the musculoskeletal system of miscellaneous endocrine disorders. Radiol Clin North Am. 1991;29(1):135-147.
pubmed - McLean RM, Podell DN. Bone and joint manifestations of hypothyroidism. Semin Arthritis Rheum. 1995;24(4):282-290.
doi pubmed - Rosenbloom AL, Silverstein JH, Lezotte DC, Richardson K, McCallum M. Limited joint mobility in childhood diabetes mellitus indicates increased risk for microvascular disease. N Engl J Med. 1981;305(4):191-194.
doi pubmed - Rosenbloom AL, Silverstein JH, Lezotte DC, Riley WJ, Maclaren NK. Limited joint mobility in diabetes mellitus of childhood: natural history and relationship to growth impairment. J Pediatr. 1982;101(5):874-878.
doi pubmed - Lindsay JR, Kennedy L, Atkinson AB, Bell PM, Carson DJ, McCance DR, Hunter SJ. Reduced prevalence of limited joint mobility in type 1 diabetes in a U.K. clinic population over a 20-year period. Diabetes Care. 2005;28(3):658-661.
doi pubmed - Francia P, Toni S, Iannone G, Seghieri G, Piccini B, Vittori A, Santosuosso U, et al. Type 1 diabetes, sport practiced, and ankle joint mobility in young patients: What is the relationship? Pediatr Diabetes. 2018;19(4):801-808.
doi pubmed - Rosenbloom AL. Limited joint mobility in childhood diabetes: discovery, description, and decline. J Clin Endocrinol Metab. 2013;98(2):466-473.
doi pubmed - Lindgren AM, Lieber AM, Shah SA, Thacker MM. Management of atypical slipped capital femoral epiphysis. J Pediatr Soc North Am. 2023;5(3):727.
doi pubmed - Clarke CF, Piesowicz AT, Spathis GS. Limited joint mobility in children and adolescents with insulin dependent diabetes mellitus. Ann Rheum Dis. 1990;49(4):236-237.
doi - Montana E, Roca A, Puche C, et al. Limited joint mobility in type 1 diabetes mellitus: relationship with microangiopathy. Diabetes Res Clin Pract. 1995;28(1):25-30.
doi - Arkkila PE, Gautier JF. Musculoskeletal disorders in diabetes mellitus: an update. Best Pract Res Clin Rheumatol. 2003;17(6):945-970.
doi pubmed - Gutch M, Kumar S, Razi SM, et al. Skeletal manifestations in juvenile hypothyroidism and response to treatment. Indian J Endocrinol Metab. 2013;17(Suppl 1):S176-S179.
doi - Lehmann CL, Arons RR, Loder RT, Vitale MG. The epidemiology of slipped capital femoral epiphysis: an update. J Pediatr Orthop. 2006;26(3):286-290.
doi - Karthikeyan K, Suresh S, Ramachandran P. Slipped capital femoral epiphysis as a presenting feature of pseudohypoparathyroidism. J Pediatr Endocrinol Metab. 2012;25(1-2):163-166.
doi - Lindgren V, Sankar WN, Novais EN, et al. Atypical slipped capital femoral epiphysis: endocrine associations and outcomes. J Pediatr Orthop. 2023;43(1):e1-e7.
doi - Dons RF, Rosselet P, Fischer JA. Arthropathy in acromegaly: long-term clinical and radiological follow-up. Ann Rheum Dis. 1988;47(8):633-640.
doi - Pelsma ICM, Biermasz NR, van Furth WR, Pereira AM, Kroon HM, Kloppenburg M, Claessen K. Progression of acromegalic arthropathy in long-term controlled acromegaly patients: 9 years of longitudinal follow-up. J Clin Endocrinol Metab. 2021;106(1):188-200.
doi pubmed - Pereira RM, Lima JG, Martins MR. Thyroid acropachy in an adolescent with Graves disease. J Pediatr Endocrinol Metab. 2019;32(6):657-661.
doi - Benina AR, Kolodkina AA, Tiul'pakov AN, et al. Primary hyperparathyroidism in children. Probl Endokrinol (Mosk). 2023;70(3):74-82.
doi - Lodish MB, Gourgari E, Sinaii N, et al. Skeletal maturation in children with Cushing syndrome is not consistently delayed: the role of corticotropin, obesity, and steroid hormones, and the effect of surgical cure. J Pediatr. 2014;164(4):801-806.
doi - Ploegstra WM, Boontje RP, Kamps AW. Arthritis associated with antithyroid therapy in a 15-year-old girl. J Pediatr Pharmacol Ther. 2011;16(2):98-101.
doi pubmed - Leong GM, Abad V, Charmandari E, Reynolds JC, Hill S, Chrousos GP, Nieman LK. Effects of child- and adolescent-onset endogenous Cushing syndrome on bone mass, body composition, and growth: a 7-year prospective study into young adulthood. J Bone Miner Res. 2007;22(1):110-118.
doi pubmed - Traisman HS, Traisman ES, Marr TJ, Wise J. Joint contractures in patients with juvenile diabetes and their siblings. Diabetes Care. 1978;1(6):360-361.
doi pubmed - Campbell RR, Hawkins SJ, Maddison PJ, Reckless JP. Limited joint mobility in diabetes mellitus. Ann Rheum Dis. 1985;44(2):93-97.
doi pubmed - Moyer J, Jacks L, Hunter JD, Chan G. Slipped capital femoral epiphysis and associated hypothyroidism. A review of the literature with two classic case examples. J Pediatr Endocrinol Metab. 2016;29(4):427-434.
doi pubmed - Nihei H, Tada H, Naruse Y, Izawa M, Kato M, Okuno H, Nakamura A, et al. Polyarthritis caused by methimazole in two Japanese patients with graves' disease. J Clin Res Pediatr Endocrinol. 2013;5(4):270-272.
doi pubmed - Tullu MS, Udgirkar VS, Muranjan MN, Sathe SA, Kamat JR. Kocher-Debre-Semelaigne syndrome: hypothyroidism with muscle pseudohypertrophy. Indian J Pediatr. 2003;70(8):671-673.
doi pubmed - Hwang S, Lee YJ, Song MH, Cho TJ, Choi IH, Shin CH. Incidence and clinical characteristics of slipped capital femoral epiphysis in patients with endocrinopathy: a population-based cohort study. J Bone Joint Surg Am. 2024;106(5):381-388.
doi pubmed - Rostomyan L, Daly AF, Petrossians P, Nachev E, Lila AR, Lecoq AL, Lecumberri B, et al. Clinical and genetic characterization of pituitary gigantism: an international collaborative study in 208 patients. Endocr Relat Cancer. 2015;22(5):745-757.
doi pubmed - Beckers A, Petrossians P, Hanson J, Daly AF. The causes and consequences of pituitary gigantism. Nat Rev Endocrinol. 2018;14(12):705-720.
doi pubmed - Claessen K, Canete AN, de Bruin PW, Pereira AM, Kloppenburg M, Kroon HM, Biermasz NR. Acromegalic arthropathy in various stages of the disease: an MRI study. Eur J Endocrinol. 2017;176(6):779-790.
doi pubmed - Colao A, Grasso LFS, Giustina A, Melmed S, Chanson P, Pereira A, Pivonello R. Acromegaly. Nat Rev Dis Primers. 2019;5(1):20.
doi - Blethen SL, Rundle AC. Slipped capital femoral epiphysis in children treated with growth hormone. A summary of the National Cooperative Growth Study experience. Horm Res. 1996;46(3):113-116.
doi pubmed - Darendeliler F, Karagiannis G, Wilton P. Headache, idiopathic intracranial hypertension and slipped capital femoral epiphysis during growth hormone treatment: a safety update from the KIGS database. Horm Res. 2007;68(Suppl 5):41-47.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, including commercial use, provided the original work is properly cited.
International Journal of Clinical Pediatrics is published by Elmer Press Inc.