| International Journal of Clinical Pediatrics, ISSN 1927-1255 print, 1927-1263 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, Int J Clin Pediatr and Elmer Press Inc |
| Journal website https://ijcp.elmerpub.com |
Original Article
Volume 15, Number 1, March 2026, pages 8-15
Efficacy of Teduglutide for Pediatric and Short Bowel Syndrome-Associated Intestinal Failure: A Systematic Review
Matheus Barros de Albuquerquea, Carla Montenegro Diasb, f , Diogo Grassano Melo Ferreiraa, Joao Victor de Oliveira Maldonadoc, Mateus Macedo Margatoc, Luiz Eduardo Duarte Borges Nunesa, Emanuel Savio Cavalcanti Sarinhob, Lucia Helena Oliveira Cordeirod, Luiz Alberto Reis Mattos Juniorb, Amanda Catriona Fifie
aDepartment of Medicine, Centro de Ciencias Medicas, Faculdade de Medicina do Recife, Universidade Federal de Pernambuco, Recife, Brazil
bDepartment of Pediatrics, Centro de Ciencias Medicas, Faculdade de Medicina do Recife, Universidade Federal de Pernambuco, Recife, Brazil
cDepartment of Medicine, Centro de Ciencias Medicas, Universidade de Brasilia, Brasilia, Distrito Federal, Brazil
dDepartment of Clinical Medicine, Centro de Ciencias Medicas, Faculdade de Medicina do Recife, Universidade Federal de Pernambuco, Recife, Brazil
eDepartment of Pediatric Gastroenterology, Hepatology and Nutrition, University of Miami, Miller School of Medicine, Miami, FL, USA
fCorresponding Author: Carla Montenegro Dias, Department of Pediatrics, Centro de Ciencias Medicas, Faculdade de Medicina do Recife, Universidade Federal de Pernambuco, Recife, Brazil
Manuscript submitted December 20, 2025, accepted March 16, 2026, published online March 26, 2026
Short title: Teduglutide for Pediatric SBS
doi: https://doi.org/10.14740/ijcp1039
| Abstract | ▴Top |
Background: Intestinal failure secondary to short bowel syndrome (SBS) is a severe malabsorptive condition often requiring long-term parenteral nutrition (PN). Teduglutide, a glucagon-like peptide-2 (GLP-2) analogue, promotes intestinal adaptation and may reduce PN dependence. This systematic review evaluated the efficacy of teduglutide in reducing PN requirements in pediatric patients with intestinal failure.
Methods: Clinical studies evaluating teduglutide therapy in pediatric patients with intestinal failure secondary to SBS were included. Randomized trials, non-randomized studies, and post-hoc analysis of clinical trials were considered due to the limited number of pediatric studies available. Risk of bias was assessed using the RoB-2 (Cochrane) tool, and statistical analyses were performed using Review Manager 5.4.1.
Results: Teduglutide therapy was associated with a significantly higher likelihood of achieving a clinically meaningful ≥ 20% reduction in PN volume compared with standard of care (odds ratio 11.79; 95% confidence interval, 2.04–68.24; P = 0.006). Treatment was also associated with improvement in length/height-for-age z-score, while no significant difference was observed in weight-for-length z-score.
Conclusion: Findings support teduglutide as an effective therapeutic option to reduce PN dependence and promote progression toward enteral nutrition autonomy in pediatric patients with SBS-associated intestinal failure.
Keywords: GLP-2; Intestinal failure; Children; Teduglutide; Pediatric; Short bowel syndrome; Parenteral nutrition
| Introduction | ▴Top |
Short bowel syndrome (SBS) results from extensive intestinal resection and leads to impaired absorption of nutrients, fluids, and electrolytes, frequently progressing to intestinal failure in pediatric patients [1]. Because adequate nutrition is essential for growth and development, children with SBS often require prolonged parenteral nutrition (PN), which is associated with significant morbidity, including infections and intestinal failure–associated liver disease [2, 3]. Reducing dependence on PN while promoting intestinal adaptation remains a major therapeutic goal in this population.
Glucagon-like peptide-2 (GLP-2) is an intestinotrophic hormone involved in promoting mucosal growth and enhancing absorptive capacity in the remaining bowel [4]. Teduglutide, a recombinant GLP-2 analogue, has been developed to stimulate intestinal adaptation and reduce PN requirements in patients with SBS [5]. Clinical guidelines support its use in adults with SBS-dependent intestinal failure [6]; however, evidence in pediatric populations is more limited.
Available pediatric studies evaluating teduglutide have reported variable outcomes, with some demonstrating improvements in enteral autonomy and growth parameters [7–9], while others highlight modest benefits and concerns regarding long-term safety [10]. The small sample sizes and heterogeneity of these studies make it difficult to draw definitive conclusions regarding the efficacy of teduglutide in children with SBS-associated intestinal failure (SBS-IF).
Given these uncertainties, we conducted a systematic review with pooled analysis. Due to the limited number of randomized controlled trials (RCTs) in pediatric SBS, this review includes randomized trials, open-label studies, and post-hoc analyses evaluating teduglutide therapy. In this study, we evaluate the efficacy of teduglutide in reducing PN requirements and improving anthropometric outcomes in pediatric patients with SBS-IF.
| Materials and Methods | ▴Top |
Data registration, conduction of the systematic review, and reporting were compliant with the Cochrane Collaboration Handbook for Systematic Review of Interventions and the Preferred Reporting Items for Systematic Reviews and Meta-analysis (PRISMA) statement guidelines [11, 12]. This systematic review was pre-registered in PROSPERO (registration number CRD420251232892) [13]. This study is a systematic review of published data and did not involve human participants directly. Therefore, institutional review board approval and informed consent were not required.
Eligibility criteria
Studies were included in this systematic review if they met the following eligibility criteria: 1) clinical studies evaluating teduglutide treatment; 2) studies including pediatric patients younger than 18 years with a diagnosis of SBS; and 3) studies reporting clinical outcomes related to PN reduction or intestinal adaptation. Randomized and non-randomized clinical studies, including open-label trials and post-hoc analyses of clinical trials, were considered eligible due to the limited number of pediatric studies available in this field.
Studies were excluded if they: 1) did not report outcomes of interest; 2) included patients without SBS; or 3) were available only as conference abstracts, posters, or unpublished protocols. When multiple publications reported outcomes from the same or overlapping clinical trial population, they were reviewed together and included only if they contributed distinct outcomes relevant to the prespecified analyses. In such cases, overlap was considered during interpretation, and the total number of participants reported across publications was not assumed to represent a fully unique population.
Search strategy and data extraction
We systematically searched for clinical studies meeting the eligibility criteria in MEDLINE, EMBASE, and Cochrane Central Register of Controlled Trials databases, from inception to July 25, 2024.
The search strategy consisted of: (Glepaglutide or Teduglutide or Apraglutide or “glucagon-like peptide 2” or “glucagon-like peptide 2” or “glucagon-like peptide-2” or GLP2 or GLP-2 or “GLP 2” or gattex OR revestive) and (“Bowel Syndrome, Short” or “Short Bowel Syndrome” or “Syndrome, Short Bowel” or “short gut” or “intestinal failure” or “short intestine syndrome” or “bowel resection”) and (“pediatric” or “paediatric” or “children” or “adolescent”).
The references from all included studies, previous systematic reviews, and meta-analysis were also searched manually for eligible studies. Gray literature was not searched. The search was independently conducted by three different authors (MBA, DGMF, and CMD), who also independently extracted the data following predefined criteria for search and quality assessment. Disagreements were resolved through consensus.
Endpoints
Outcomes of interest included: 1) the proportion of patients achieving a clinically meaningful reduction of ≥ 20% in parenteral support (PS) volume at 24 weeks, defined as the primary outcome; 2) mean change in PS volume at the teduglutide dose of 0.05 mg/kg; 3) mean change in PS volume at the teduglutide dose of 0.025 mg/kg; 4) mean change in weight-for-length z-score; and 5) mean change in length/height-for-age z-score.
Quality assessment
The risk of bias of each study was evaluated using Cochrane Risk of Bias Tool for Randomized Trials 2 (RoB-2) [14]. Two independent authors conducted a risk assessment (JVOM and MMM). Disagreements were resolved through consensus after discussing reasons for discrepancy. Grading of Recommendations, Assessment, Development, and Evaluations (GRADE) pro software (Evidence Prime) was used by two independent reviewers to assess the quality of evidence for each outcome across the included studies. GRADE criteria were used during the evaluation process [15].
Statistical analysis
Odds ratio (OR) was computed using the Mantel-Haenszel test for binary outcomes and mean difference (MD) for continuous data with 95% confidence interval (CI) applied as a measure of effect size. It was considered P-values of < 0.05 to be statistically significant for the primary outcome. To evaluate heterogeneity, Cochran Q test and I2 statistics were used. I2 values < 10% were considered negligible for heterogeneity, 10% to 39% low, 40% to 59% moderate, 60% to 89% high, and ≥ 90% very high [16].
To address possible variations in clinical and methodological aspects among the studies, DerSimonian and Laird random-effects models were applied in all outcomes, regardless of I2 result for heterogeneity [16]. Review Manager 5.4 (Nordic Cochrane Centre, The Cochrane Collaboration) was used for statistical analysis.
| Results | ▴Top |
Study selection and baseline characteristics
The systematic search yielded 457 potential studies as detailed in Figure 1. After removing duplicates and studies that did not meet our inclusion criteria on title or abstract, 44 articles were fully assessed. Finally, after meticulous assessment, three studies were included [7–9]. The included publications reported data on 172 participants overall, of whom 144 received teduglutide; however, because some reports were post-hoc or secondary analyses of previously described trial populations, partial overlap of participants across publications could not be completely excluded.
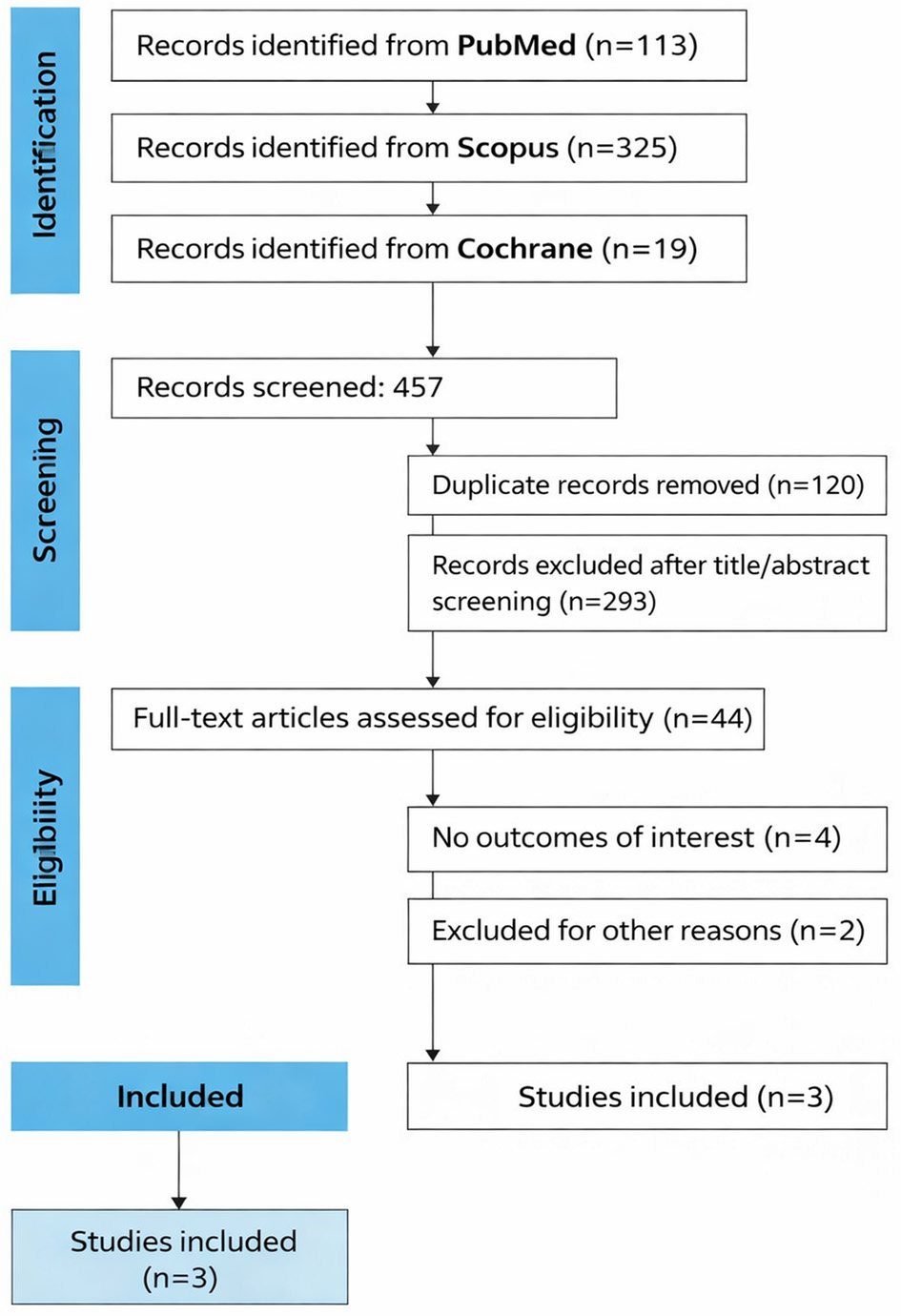 Click for large image | Figure 1. Study selection for systematic review. |
The mean (SD) weight-for-age z-score ranged from −1.5 to −0.2 on patients receiving teduglutide and from −0.6 to 0.2 on patients receiving standard of care (SoC) treatment. Individual study characteristics are summarized in Table 1.
 Click to view | Table 1. Summary of Individual Study Characteristics |
Primary outcome (PS 0.05 mg/kg)
The primary clinically relevant outcome in the included studies was the proportion of patients achieving a ≥ 20% reduction in PS volume after 24 weeks of treatment. Teduglutide was associated with a significantly higher proportion of patients achieving this endpoint compared with SoC (OR, 11.79; 95% CI, 2.04–68.24; P = 0.006; I2 = 0%; Fig. 2).
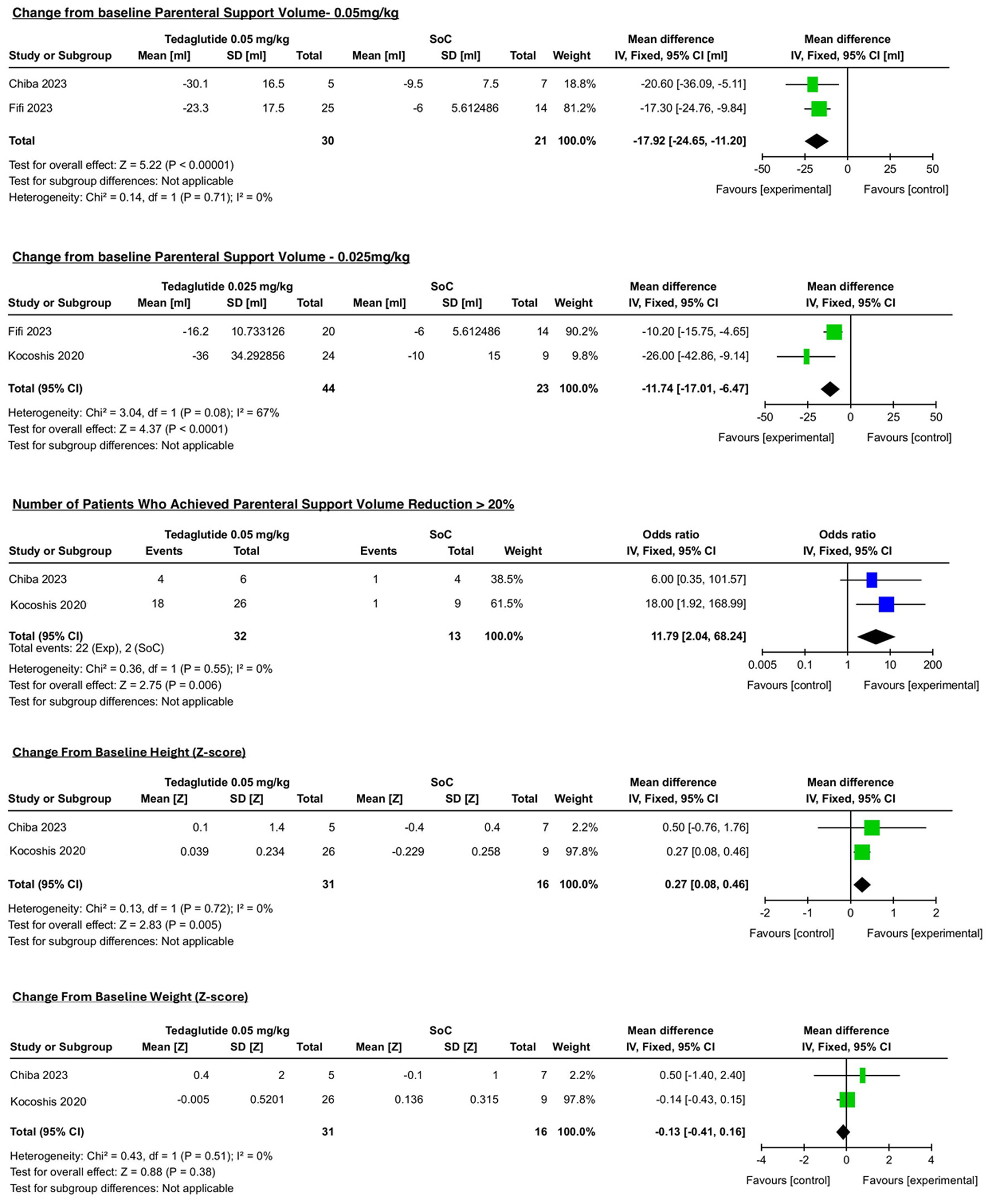 Click for large image | Figure 2. Outcomes. |
In addition, pooled analysis showed that teduglutide significantly reduced PS volume at the 0.05 mg/kg dose compared with SoC (MD, −17.92 mL/kg/day; 95% CI, −24.65 to −11.20; P < 0.00001; I2 = 0%; Fig. 2).
Functional outcomes
Teduglutide was also associated with a significant reduction in PS volume at the 0.025 mg/kg dose compared with SoC (MD, −11.74 mL/kg/day; 95% CI, −17.01 to −6.47; P < 0.00001; I2 = 67%; Fig. 2).
Across the included studies, a greater proportion of patients receiving teduglutide achieved a clinically meaningful reduction in PS requirements (≥ 20% reduction), supporting the functional benefit of teduglutide in reducing dependence on PN in pediatric SBS-IF.
Anthropometric outcomes (height and weight)
We found statistical significance for z-score change to baseline for length/height-for-age (MD, 0.27; 95% CI, 0.08 to 0.46; P = 0.005; I2 = 0%; Fig. 2). In contrast, the weight-for-length z-score, mean change (MD, −0.13; 95% CI, −0.41 to 0.16; P = 0.38; I2 = 0%; Fig. 2) showed no statistically significant difference between groups.
Quality assessment
The RoB-2 tool was used to assess the quality of included studies. A summary of the risk of bias assessment is shown in Table 2. Fifi (2023) and Chiba (2023) studies were rated at an overall moderate risk of bias, and Kocoshis (2020) was rated at an overall low risk of bias. According to the GRADE tool, the overall certainty of the evidence was moderate for the main outcome and for the secondary outcomes, regarding the risk of bias effect, inconsistency, imprecision, and publication bias.
 Click to view | Table 2. Summary of the Risk of Bias Assessment |
| Discussion | ▴Top |
This systematic review included three clinical studies evaluating teduglutide in pediatric patients with SBS-IF, encompassing both randomized and non-randomized study designs. Across the studies included, teduglutide therapy was associated with clinically meaningful reductions in PS requirements compared with baseline SoC management.
Baseline anatomical characteristics were reported across the included studies and were generally comparable between treatment groups. Residual small bowel length ranged from approximately 1.5 to 95 cm, and most patients had colon-in-continuity and preserved ileocecal valve when reported. The most common underlying etiologies of SBS included gastroschisis, necrotizing enterocolitis, and midgut volvulus.
Importantly, a significantly higher proportion of patients in the teduglutide group achieved a ≥ 20% reduction in PS volume compared to controls. Changes in length/height-for-age z-score were also statistically significant between groups, while no significant difference was observed in weight-for-length z-score mean change. These findings indicate that teduglutide contributes to meaningful improvements in nutritional support requirements and selected growth parameters in pediatric patients with SBS-IF.
Kocoshis et al (2020) showed that the number of patients who achieved a 20% reduction in PS from baseline at week 24 was achieved by 13 (54.2%), 18 (69.2%), and one (11.1%) patients who received 0.025 mg/kg teduglutide, 0.05 mg/kg teduglutide, and SoC, respectively (P < 0.05 vs. SoC) demonstrating that teduglutide significantly reduced PS volume at both 0.05 and 0.025 mg/kg doses [7].
However, the number of patients receiving the lower dose was limited. Therefore, although reductions in PS requirements were reported at this dosage, definitive conclusions regarding the efficacy of the 0.025 mg/kg dose cannot be established based on the currently available evidence. Future studies with larger pediatric cohorts are needed to better determine the effectiveness of lower dosing strategies.
The reduction in PS requirements observed in this systematic review is particularly important given the well-recognized risks of long-term PN, including infectious complications and intestinal failure-associated liver disease [17].
These results are consistent with previous studies suggesting that teduglutide promotes intestinal adaptation and supports enteral autonomy in pediatric SBS-IF [18].
A notable finding of this analysis was the significantly higher number of patients achieving a ≥ 20% reduction in PS volume over a 24-week period in the teduglutide group. This outcome reflects not only a decrease in PS volume but also a clinically meaningful step toward increased nutritional independence.
Across the included studies, enteral autonomy was achieved in seven of 58 pediatric patients (12.1%) including five patients in the study by Kocoshis et al and two children in the study by Chiba et al. Enteral autonomy outcomes were not reported in the post-hoc analysis by Fifi et al and therefore were not included in this calculation.
Similar improvements in PS reduction and enteral autonomy have been reported in prior pediatric studies evaluating teduglutide therapy [19], supporting the robustness of the findings observed in the pooled analysis. In 2023, a single-center study observed enteral autonomy after teduglutide use within 48 weeks for 32% of children and a significant reduction in PN requirements was observed in the other pediatric patients who did not achieve complete enteral autonomy [20]. Similarly, in 2025, a trial reported achievement of 30% of pediatric patients with SBS achieved enteral autonomy at 71.57 weeks of treatment with the GLP-2 analogue and a decrease in PS volume of ≥ 20% experienced by 67% of 27 pediatric patients [21].
Anthropometric outcomes demonstrated a differential response, with a significant improvement in linear growth but no corresponding statistically significant change in weight. This pattern may reflect improved nutrient absorption and intestinal adaptation associated with teduglutide therapy, which could contribute to linear growth over time [22]. The absence of a significant effect on weight gain may reflect the complex interplay of nutritional intake, disease severity, and ongoing dependence on PS in this population [23].
As results may vary in children with SBS treated with teduglutide and considering the high cost of the medication, there is a clear need for further studies on when to indicate the GLP-2 agonist and who are the most beneficiated patients. In 2025, a real-life multicenter cohort study observed that longer residual small bowel, better nutritional status, and absence of liver disease were associated with response to teduglutide. Complete PN weaning was predicted by lower calories needs and higher citrulline at baseline. Increases of hemoglobin and citrulline in the first 6 months of treatment were further predictors of complete PN weaning. Even though the study was limited by its open label design, these findings may guide a tailored indication for the use of teduglutide in children with SBS [24].
In a similar way, a retrospective single-center analysis published in 2025 suggested that pediatric patients older than 5 years and with less dependency indexes of PS experienced more significant decrease in PS volume compared to younger children or patients with higher dependency of PS [21].
Teduglutide treatment was generally well tolerated. The most frequently reported adverse events included vomiting, abdominal pain, diarrhea, pyrexia, and upper respiratory infections, with most events described as mild to moderate in severity. No unexpected safety signals were reported.
Serious adverse events were infrequently reported across the included studies. In the phase III trial by Kocoshis et al, two serious gastrointestinal adverse events were reported: gastrointestinal obstruction with thickened stool and adynamic ileus. Both events required hospitalization, were considered moderate in severity, resolved after temporary interruption of teduglutide, and did not recur after treatment was resumed. No deaths were reported during the study.
In the study by Chiba et al, several treatment-emergent serious adverse events were observed during the study period; however, only one serious adverse event—abdominal pain—was considered related to teduglutide treatment. No deaths occurred.
In the post-hoc analysis by Fifi et al, adverse events were predominantly gastrointestinal symptoms such as vomiting, abdominal pain, diarrhea, and nausea, and most were classified as mild or moderate. No adverse events leading to treatment discontinuation or death were reported.
To sum up, the findings were encouraging. However, several limitations must be considered when interpreting the results of this systematic review. The small number of included studies and the limited sample size reduce the statistical power and generalizability of the conclusions. Additionally, heterogeneity in study design, patient characteristics, and outcome definitions may have influenced the pooled estimates. Furthermore, some included studies consisted of pooled or post-hoc analyses of previously conducted clinical trials, and therefore partial overlap of patient populations across studies cannot be completely excluded. These limitations highlight the need for future randomized trials with standardized outcome measures and larger pediatric prospective clinical trials.
It should also be noted that the studies included in this analysis primarily evaluated short-term outcomes. Therefore, long-term safety, durability of PS reduction, and sustained impact on growth remain uncertain. Longitudinal studies are needed to determine whether the benefits observed with teduglutide are maintained over extended treatment periods and to better characterize potential long-term adverse effects.
Overall, the available evidence suggests that teduglutide represents a promising therapeutic option for reducing PS requirements and promoting intestinal adaptation in pediatric SBS-IF. Nevertheless, larger prospective studies with longer follow-up are required to better define optimal dosing strategies, patient selection, and long-term safety.
Acknowledgments
The authors used ChatGPT (OpenAI) to assist with language editing and text organization. The authors take full responsibility for the content, interpretation of the data, and conclusions presented in this manuscript.
Financial Disclosure
This study received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors declare that they have no conflict of interest related to this manuscript.
Informed Consent
Not applicable.
Author Contributions
Matheus Barros de Albuquerque: conceptualization, methodology, software, writing – original draft, project administration. Diogo Grassano Melo Ferreira: data analysis, software, writing – original draft. Joao Victor de Oliveira Maldonado: methodology, software, writing. Mateus Macedo Margato: methodology, software, writing. Carla Montenegro Dias: writing, supervision, reviewing, publishing. Luiz Eduardo Duarte Borges Nunes: Methodology, data curation. Emanuel Savio Cavalcanti Sarinho: project visualization, data curation. Lucia Helena Oliveira Cordeiro: project visualization, formal analysis. Luiz Alberto Reis Mattos Junior: writing, reviewing. Amanda Catriona Fifi: project visualization, formal analysis, supervision.
Data Availability
All data analyzed in this study are derived from previously published studies. The data supporting the findings of this study are available within the included articles and from the corresponding author upon reasonable request.
Abbreviations
CI: confidence interval; EMBASE: Excepta Medica database; GLP-2: glucagon-like peptide-2; GRADE: Grading of Recommendations, Assessment, Development, and Evaluation; IF: intestinal failure; MD: mean difference; MEDLINE: Medical Literature Analysis and Retrieval System Online; OR: odds ratio; PN: parenteral nutrition; PRISMA: preferred reporting items for systematic review and meta-analysis; PS: parenteral support; RCTs: randomized controlled trials; RevMan: Review Manager; RoB-2: Risk of Bias 2; SBS: short bowel syndrome; SD: standard deviation; SoC: standard of care
| References | ▴Top |
- Massironi S, Cavalcoli F, Rausa E, Invernizzi P, Braga M, Vecchi M. Understanding short bowel syndrome: Current status and future perspectives. Dig Liver Dis. 2020;52(3):253-261.
doi pubmed - Uko V, Radhakrishnan K, Alkhouri N. Short bowel syndrome in children: current and potential therapies. Paediatr Drugs. 2012;14(3):179-188.
doi pubmed - Caporilli C, Gianni G, Grassi F, Esposito S. An overview of short-bowel syndrome in pediatric patients: focus on clinical management and prevention of complications. Nutrients. 2023;15(10):2341.
doi pubmed - Rosete BE, Wendel D, Horslen SP. Teduglutide for pediatric short bowel syndrome patients. Expert Rev Gastroenterol Hepatol. 2021;15(7):727-733.
doi pubmed - Wales PW, Hill S, Robinson I, Raphael BP, Matthews C, Cohran V, Carter B, et al. Long-term teduglutide associated with improved response in pediatric short bowel syndrome-associated intestinal failure. J Pediatr Gastroenterol Nutr. 2024;79(2):290-300.
doi pubmed - Pironi L, Cuerda C, Jeppesen PB, Joly F, Jonkers C, Krznaric Z, Lal S, et al. ESPEN guideline on chronic intestinal failure in adults - Update 2023. Clin Nutr. 2023;42(10):1940-2021.
doi pubmed - Kocoshis SA, Merritt RJ, Hill S, Protheroe S, Carter BA, Horslen S, Hu S, et al. Safety and efficacy of teduglutide in pediatric patients with intestinal failure due to short bowel syndrome: a 24-week, phase III study. JPEN J Parenter Enteral Nutr. 2020;44(4):621-631.
doi pubmed - Fifi A, Raphael BP, Terreri B, Uddin S, Kaufman SS. Effects of teduglutide on diarrhea in pediatric patients with short bowel syndrome-associated intestinal failure. J Pediatr Gastroenterol Nutr. 2023;77(5):666-671.
doi pubmed - Chiba M, Masumoto K, Kaji T, Matsuura T, Morii M, Fagbemi A, Hill S, et al. Efficacy and safety of teduglutide in infants and children with short bowel syndrome dependent on parenteral support. J Pediatr Gastroenterol Nutr. 2023;77(3):339-346.
doi pubmed - Kim DW, Kim E, Bertram K, Rim DS, Nolen-Doerr E, Shin JH. Long-term outcomes and adverse effects of teduglutide in patients with short bowel syndrome: Highlighting hyperamylasemia and hyperlipasemia. Am J Health Syst Pharm. 2024;81(4):146-152.
doi pubmed - Higgins JPT, Thomas J, Chandler J, et al. eds. Cochrane handbook for systematic reviews of interventions. Version 6.4. Cochrane; 2023. Updated August 2023. www.training. cochrane.org/handbook.
- Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, Shamseer L, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71.
doi pubmed - https://www.crd.york.ac.uk/PROSPERO/display_record.php?ID=CRD420251232892.
- Sterne JAC, Savovic J, Page MJ, Elbers RG, Blencowe NS, Boutron I, Cates CJ, et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366:l4898.
doi pubmed - Balshem H, Helfand M, Schunemann HJ, Oxman AD, Kunz R, Brozek J, Vist GE, et al. GRADE guidelines: 3. Rating the quality of evidence. J Clin Epidemiol. 2011;64(4):401-406.
doi pubmed - Schober P, Mascha EJ, Vetter TR. Statistics from A (Agreement) to Z (z Score): a guide to interpreting common measures of association, agreement, diagnostic accuracy, effect size, heterogeneity, and reliability in medical research. Anesth Analg. 2021;133(6):1633-1641.
doi pubmed - Kim ES, Keam SJ. Teduglutide: A Review in Short Bowel Syndrome. Drugs. 2017;77(3):345-352.
doi pubmed - Pulido AV, Kellogg K, Hunt K, Davis C. Development of a clinical practice guideline for weaning and discontinuing parenteral nutrition in hospitalized children as part of a central line-associated bloodstream infection-focused quality improvement initiative. JPEN J Parenter Enteral Nutr. 2021;45(8):1653-1662.
doi pubmed - Seidner DL, Gabe SM, Lee HM, Olivier C, Jeppesen PB. Enteral autonomy and days off parenteral support with teduglutide treatment for short bowel syndrome in the STEPS trials. JPEN J Parenter Enteral Nutr. 2020;44(4):697-702.
doi pubmed - Lambe C, Talbotec C, Kapel N, Barbot-Trystram L, Brabant S, Nader EA, Pigneur B, et al. Long-term treatment with teduglutide: a 48-week open-label single-center clinical trial in children with short bowel syndrome. Am J Clin Nutr. 2023;117(6):1152-1163.
doi pubmed - Josey C, Mazariegos G, King E, Holzer P, Rudolph J, Raghu VK. Real-world efficacy of long-term teduglutide use in pediatric patients with short bowel syndrome. Intest Fail. 2025;7.
doi pubmed - Gombosova L, Suchansky M, Krivus J, Hornova J, Havlicekova Z, Fojtova A, Norek B, et al. Evaluation of the effectiveness of teduglutide treatment in patients with short bowel syndrome in Slovakia-multicenter real-world study. J Clin Med. 2024;13(5):1238.
doi pubmed - Ishida M, Kanamori Y, Hori N, Inaoka T, Ohtsuka E. In vitro and in vivo activities of T4 endonuclease V mutants altered in the C-terminal aromatic region. Biochemistry. 1990;29(16):3817-3821.
doi pubmed - Norsa L, Ghirardi A, Ramos Boluda E, Guz-Mark A, Hojsak I, Hilberath J, Broekaert IJ, et al. Predictors of response and enteral autonomy in children with short bowel syndrome treated with teduglutide: a real-life multicentre cohort study. EClinicalMedicine. 2025;85:103343.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, including commercial use, provided the original work is properly cited.
International Journal of Clinical Pediatrics is published by Elmer Press Inc.