| International Journal of Clinical Pediatrics, ISSN 1927-1255 print, 1927-1263 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, Int J Clin Pediatr and Elmer Press Inc |
| Journal website https://ijcp.elmerpub.com |
Original Article
Volume 15, Number 1, March 2026, pages 26-30
Significance of Nucleated Red Blood Cells as a Predictor of Clinical Outcome of Asphyxia in Neonates
Dayinta Rahma Ardiyantia, Nyoman Suci Widyastitib, e, Moh Syarofil Anamc, Johan Arifind
aClinical Pathology Residency Program, Faculty of Medicine, Universitas Diponegoro, Semarang, Indonesia
bNyoman Suci Widyastiti, Department of Clinical Pathology, Faculty of Medicine, Universitas Diponegoro/Clinical Pathology Division, Department of Supporting Medicine, dr Kariadi General Hospital, Semarang, Indonesia
cDepartment of Pediatrics, Faculty of Medicine, Universitas Diponegoro, Semarang, Indonesia
dDivision of Pediatric Anesthesia, Faculty of Medicine, Universitas Diponegoro/Dr. Kariadi General Hospital, Semarang, Indonesia
eCorresponding Author: Nyoman Suci Widyastiti, Nyoman Suci Widyastiti, Department of Clinical Pathology, Faculty of Medicine, Universitas Diponegoro/Clinical Pathology Division, Department of Supporting Medicine, dr Kariadi General Hospital, Semarang, Indonesia
Manuscript submitted November 23, 2025, accepted January 21, 2026, published online March 26, 2026
Short title: NRBCs and Neonatal Asphyxia Outcome
doi: https://doi.org/10.14740/ijcp1036
| Abstract | ▴Top |
Background: Neonatal asphyxia is a leading cause of premature death and a significant concern in Indonesia. An increased number of nucleated red blood cells (NRBCs) at birth reflected hypoxic conditions during late pregnancy and delivery, and functioned as a prognostic biomarker for complications related to asphyxia. NRBCs served as a prognostic biomarker in predicting complications of asphyxia. This study aimed to show that NRBCs levels could predict mortality in neonates with asphyxia.
Methods: A total of 108 neonates with asphyxia in the neonatal intensive care unit (NICU) were included in this study. Hematology examinations were carried out on the neonates, and diagnostic testing used the cutoff value of the receiver operating characteristic (ROC) curve. Furthermore, the Chi-square test and logistic regression were carried out to determine the independent relationship between NRBCs levels and mortality in neonatal asphyxia.
Results: A cutoff value of 1.28/µL was used for absolute and relative NRBCs levels, leading to a sensitivity and specificity of 83.67% and 84.75%, respectively. Furthermore, a cutoff value of 10.5% was used for relative NRBCs levels, leading to 81.63% and 81.36% as sensitivity and specificity, respectively. Multivariate analysis showed that absolute NRBCs levels influenced mortality in neonatal asphyxia, with a prevalence ratio of 10.332.
Conclusion: Absolute NRBCs levels could be considered a predictor of mortality in neonatal asphyxia.
Keywords: Nucleated red blood cells; Neonate; Asphyxia
| Introduction | ▴Top |
Neonatal asphyxia is a significant cause of neonatal morbidity and mortality worldwide. This condition, also known as perinatal asphyxia or birth asphyxia, is characterized by failure to initiate and maintain breathing at birth, which can lead to oxygen deprivation in the newborn. The World Health Organization (WHO) reported that asphyxia is responsible for approximately 900,000 neonatal deaths annually, making it a leading cause of premature mortality in neonates [1].
Based on a Sustainable Development Goal (SDG), the WHO has set a target to reduce the mortality rate of neonates. The target of reducing the mortality rate to 12 infant deaths per 1,000 live births from 25 per 1,000 is expected to be achieved by 2030 [2].
Asphyxia in neonates remains a significant concern in Indonesia, with an incidence rate of 40 per 1,000 live births. Neonatal asphyxia caused approximately 21.6% of neonatal deaths nationally in 2015 [3]. A study conducted at Dr. Soetomo General Hospital in Surabaya reported an incidence of neonatal asphyxia of 8% among newborns in 2009 [4]. Another study at RA Kartini Hospital in Jepara found that 4.1% of 148 mothers who experienced premature rupture of membranes experienced asphyxia in 2019 [5]. In contrast, the incidence of neonatal asphyxia at our institution, Dr. Kariadi Hospital, was 41.4%, with a mortality rate of 18%. These figures were higher than those reported in other regions.
The need for early identification of hypoxic exposure has led to increased attention on NRBCs, the immature red blood cells present in the fetal circulation. Increased NRBCs at birth show hypoxic conditions during late pregnancy and delivery and have been associated with perinatal asphyxia. NRBCs were released into the bloodstream in response to hypoxic stress. Elevated levels serve as a hematological marker of fetal hypoxia. Newborns with asphyxia are known to exhibit significantly higher NRBCs counts. The study found that the average NRBCs count in neonates with asphyxia was 17.43 ± 19.86 per 100 leukocytes. Meanwhile, in the control group, the average NRBCs count was 2.97 ± 4.79 per 100 leukocytes, a statistically significant difference (P < 0.0001) [6].
Other studies have reported that increased NRBCs counts are associated with adverse outcomes in neonates with asphyxia. This result suggested that NRBCs served as a prognostic biomarker in predicting complications of asphyxia [7, 8]. This low-cost prognostic biomarker is particularly needed in countries with a high prevalence of neonatal asphyxia. However, to date, no studies have evaluated the prognostic value or determined the optimal cutoff point of NRBC in such settings, particularly in Indonesia. Therefore, this study aimed to examine NRBCs levels as a predictor of clinical outcomes in neonatal asphyxia.
| Materials and Methods | ▴Top |
This analytical observational study used a cross-sectional design and was conducted from July 2024 to February 2025 at Dr. Kariadi General Hospital, Semarang. A total of 128 subjects were diagnosed with neonatal asphyxia. Inclusion criteria were neonates diagnosed with asphyxia in the neonatal intensive care unit (NICU) of Dr. Kariadi General Hospital, Semarang, and with patient medical records containing all necessary data. After 20 neonates were excluded due to the lack of hematology results, the final number of subjects was 108. The diagnosis of asphyxia was based on APGAR score assessed at the first minute of birth, with a score ≤ 7.
The diagnosis of neonatal asphyxia was classified into three categories based on the APGAR score. Mild asphyxia was defined as an APGAR score of 7–9, moderate asphyxia as a score of 4–6, and severe asphyxia as a score of 0–3. This classification was based on the National Clinical Practice Guideline on the Management of Asphyxia issued by the Ministry of Health of the Republic of Indonesia in 2019 [9].
Hematology examinations were carried out using an automated hematology analyzer (Sysmex XN-1000TM). Absolute (µL) and relative (%) NRBCs levels were obtained from the analysis results of the hematology analyzer. The clinical outcomes of asphyxia were based on patient mortality.
The accuracy of NRBCs levels with cutoff values was analyzed using diagnostic tests. The diagnostic ability of NRBCs levels was expressed as sensitivity and specificity. Further analysis used the Chi-square test and the results were considered significant when P < 0.05. Logistic regression was used to determine the independent relationship between NRBCs levels and mortality in neonatal asphyxia.
This study was reviewed and approved by the Health Research Ethics Committee of Dr. Kariadi General Hospital, Semarang, Indonesia (No. 16368/EC/KEPEK-RSDK/2025). This study was conducted in compliance with the ethical standards of the responsible institutional committee on human subjects and in accordance with the principles of the Declaration of Helsinki.
| Results | ▴Top |
A total of 108 neonates were used as subjects in this study, consisting of 59 males (54.6%) and 49 females (45.4%). Table 1 shows the demographic data for the subject population based on outcomes.
 Click to view | Table 1. Subject Demographic Data Based on Outcomes |
The test results of demographic data based on outcomes, such as birth weight, asphyxia severity, and neonates on ventilators, obtained P-values < 0.05, showing a significant relationship between these variables and outcomes. Receiving operator characteristic (ROC) testing results for absolute (Fig. 1) and relative (Fig. 2) NRBCs showed an area under the curve (AUC) of 0.859 and 0.829, with a cutoff value of 1.28/µL and 10.5%, respectively. These results are demonstrated in Table 2.
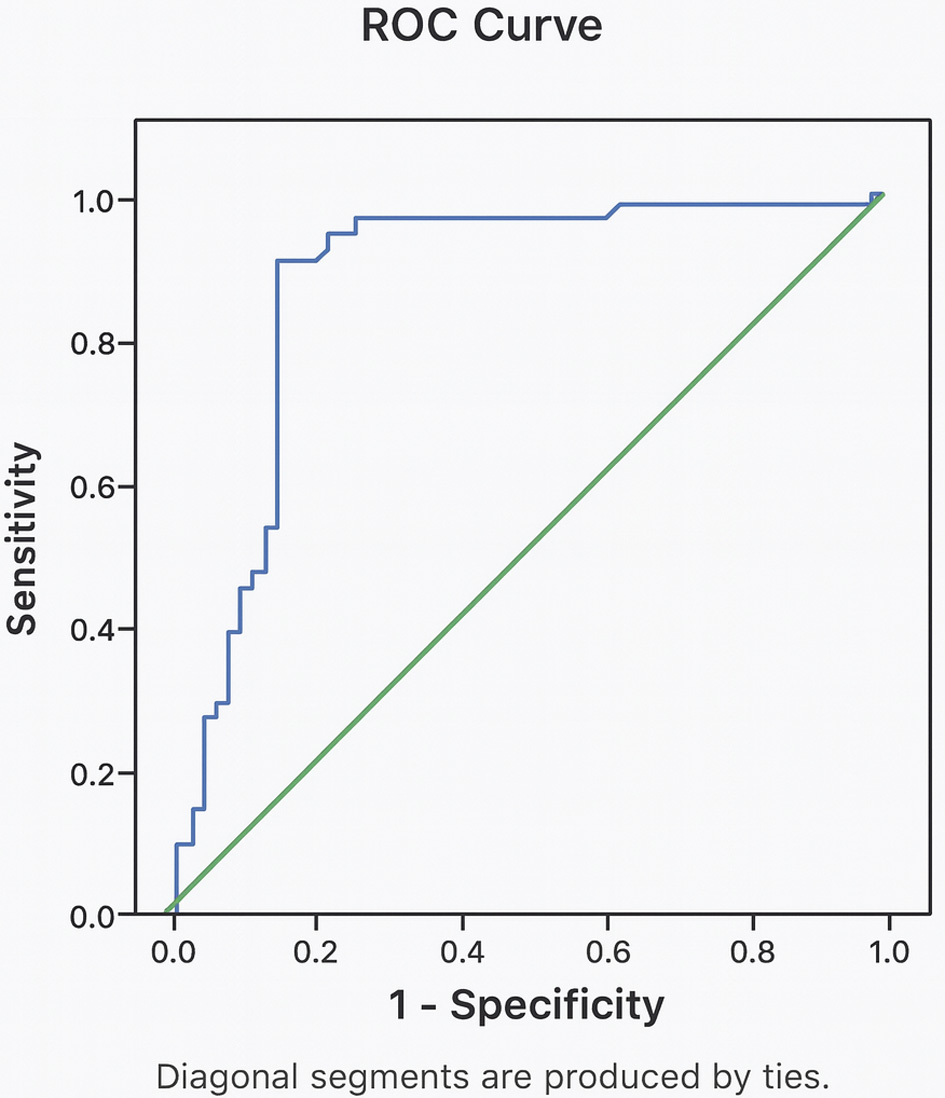 Click for large image | Figure 1. ROC curve analysis of absolute NRBC stratified by outcomes. NRBC: nucleated red blood cell; ROC: receiver operating characteristic. |
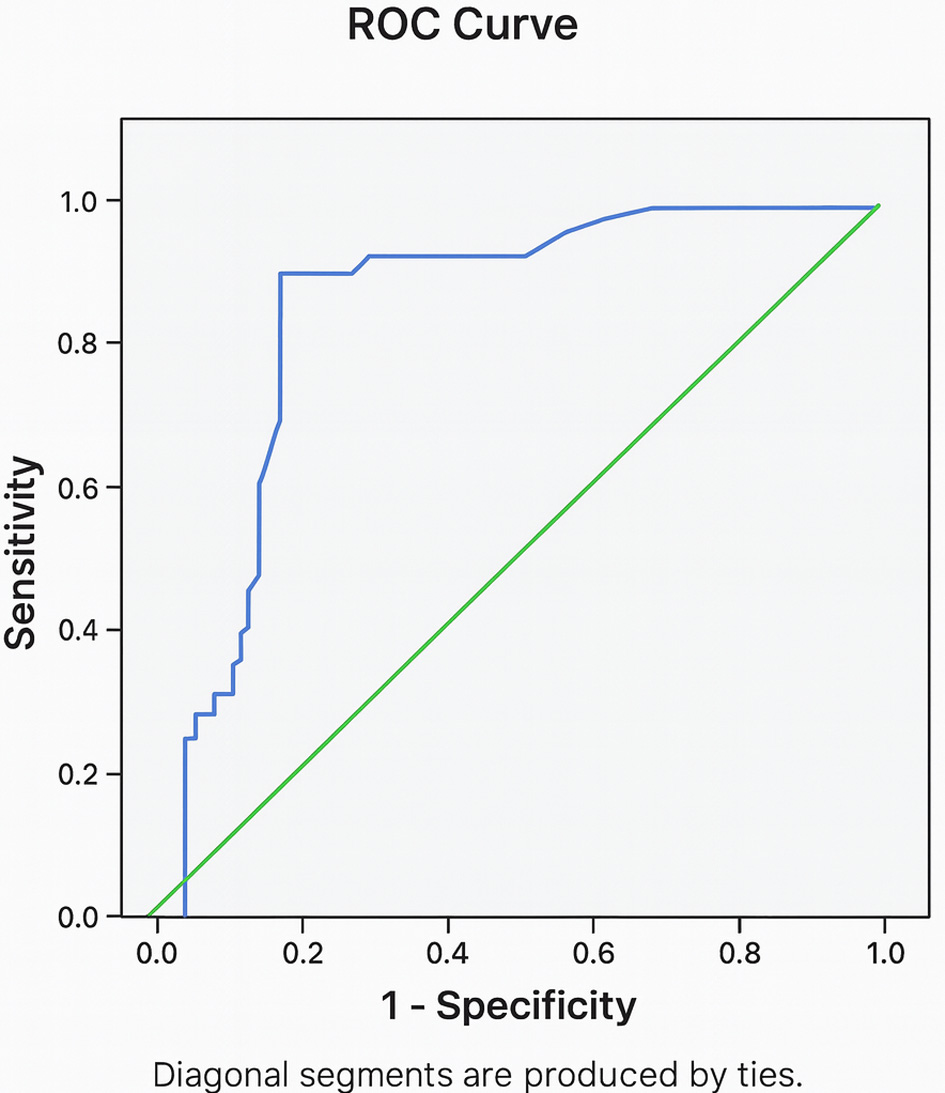 Click for large image | Figure 2. ROC curve analysis of relative NRBC stratified by outcomes. NRBC: nucleated red blood cell; ROC: receiver operating characteristic. |
 Click to view | Table 2. Absolute and Relative NRBCs Level Cutoffs Based on Outcomes |
The Chi-square test assessing the correlation between absolute NRBC counts and clinical outcomes demonstrated a statistically significant correlation, with a P-value of < 0.001 (P < 0.05), as shown in Table 3. This finding corresponded to a sensitivity of 83.67% and a specificity of 84.75%. Similarly, analysis of relative NRBC counts in relation to outcomes also revealed a significant correlation (P < 0.001), with a sensitivity of 81.63% and a specificity of 81.36%.
 Click to view | Table 3. Correlation of Absolute and Relative NRBCs to Outcomes |
Bivariate test results showed that the birth weight, severity of asphyxia, mode of delivery, and absolute and relative NRBCs met the requirements for multivariate testing. Therefore, multivariate testing of these variables was conducted and the results are demonstrated in Table 4.
 Click to view | Table 4. Results of Multivariate Tests on Outcomes |
The logistic regression test on the severity of asphyxia, absolute, and relative NRBCs obtained P-value < 0.05. Therefore, a significant relationship existed between these variables. The beta value (B) obtained a value of 2.335, suggesting that absolute NRBCs had a more dominant influence on the incidence of asphyxia mortality in neonates.
| Discussion | ▴Top |
The results of this study obtained an absolute NRBCs cutoff value of 1.28/µL with a sensitivity, specificity, and P-value of 83.67%, 84.75%, and < 0.001 (P <0.05), respectively. Relative NRBCs cutoff value was 10.5% with a sensitivity of 81.63%, specificity of 81.36%, and P-value of < 0.001 (P < 0.05). This result shows that NRBCs are statistically significantly associated with the outcomes of neonatal asphyxia (P < 0.05). The results of this study are consistent with Iqbal et al, who found that the average NRBCs count was significantly higher in the case group (16.62 ± 17.10) compared to the control group (1.69 ± 2.42) (P = 0.0001). The average neonatal NRBCs count was significantly associated with the severity and outcomes of hypoxia (P = 0.0001). According to a previous study, NRBCs levels were a hematological biomarker in newborns that was associated with intrauterine hypoxia and compensatory responses [10].
NRBCs are premature erythrocyte precursors normally present in the bone marrow of humans of all ages. These myeloid progenitor cells differentiate into more developed cells, thereby becoming erythroblasts. At this stage, the nucleus is excised, and the cell becomes a reticulocyte, developing into a mature erythrocyte. NRBCs count at birth is a biomarker of neonatal hypoxia, both in terms of its duration and its estimated severity. NRBCs levels are positively correlated with indices responsible for estimating pulmonary vascular resistance and are used as predictors of outcomes in infants with hypoxic conditions, including asphyxia [11]. The results of this study also found that birth weight, severity of asphyxia, ventilator use, and absolute and relative NRBCs levels were factors influencing outcomes (death or survival) in neonatal asphyxia (P < 0.05). Low birth weight is associated with an increased risk of poor health outcomes in childhood and adulthood [12]. Therefore, birth weight is associated with outcomes in neonatal asphyxia (P < 0.05). This result is consistent with the report of a previous study by Ardissino et al that low birth weight is a common complication in pregnancy, affecting approximately 15% of live births globally. It is also associated with a higher risk of hypoxia and cardiometabolic disease [13]. Several factors can affect blood circulation and gas exchange between the mother and fetus, leading to fetal hypoxia and neonatal asphyxia. However, there is a higher prevalence in low birth weight infants [14].
Neonates with asphyxia are classified as mild, moderate, and severe. This study found that the severity of asphyxia was associated with its outcomes (P < 0.05). The results are consistent with the report of Mietzsch et al that the severity of neonatal asphyxia is associated with morbidity and mortality in neonatal asphyxia patients [15]. Neonatal asphyxia is a life-threatening condition caused by impaired blood gas exchange, which can lead to progressive hypoxemia and hypercapnia. Severe asphyxia leads to more serious complications and a worse long-term outcome. Furthermore, severe hypoxia disrupts cellular oxygen metabolism, leading to neuronal depolarization and ischemia. This condition causes a series of disorders, ranging from reduced glucose availability needed for cellular metabolism to cell apoptosis. Asphyxia causes permanent brain damage which leads to progressive disability or death [16].
Recent data show that 5% of full-term infants require positive pressure ventilation, 2% require intubation, 0.1% require cardiac compression, and 0.05% require adrenaline. In neonatal asphyxia, optimizing resuscitation is crucial to prevent morbidity and mortality. Therefore, the primary focus of newborn resuscitation, specifically for those experiencing asphyxia, is establishing effective ventilation [17]. This study found that ventilator use is associated with outcomes in neonatal asphyxia (P < 0.05), consistent with the report of Giannakis et al. The rate of encephalopathy, lower pCO2 levels in the first 24 h after birth, and increased oxygen supplementation during the cooling period were significantly higher in the ventilator-treated group, leading to worse short-term outcomes. This result can be explained by the fact that acute hypoxic-ischemic events, as well as the reperfusion/reoxygenation phase, are characterized by increased oxidative stress initiated through the production of free radicals, leading to delayed cell death and neuronal loss [17–19].
Conclusions and suggestions
In conclusion, the results of this study show that an absolute NRBCs cutoff value of 1.28/µL has a sensitivity and specificity of 83.67% and 84.75%, respectively. The relative NRBCs with a cutoff of 10.5% also have a sensitivity and specificity of 81.63% and 81.36%, respectively. These cutoff values could be applied in routine clinical practice, particularly in developing countries with a high incidence of neonatal asphyxia, such as Indonesia. Multivariate analysis shows that absolute NRBCs levels are a predictor of mortality in neonates with asphyxia, and prevalence ratio is 10.332.
The limitation of this study includes a small sample size and a single-center design. Other factors, such as birth weight, severity of asphyxia, ventilator use, and maternal-perinatal variabels (including a history of infection, preeclamsia, intrauterine growth restriction) should be considered in assessing the prognosis of infants with neonatal asphyxia to enable more comprehensive interventions. In addition, this study evaluated only mortality as an outcome. Future studies should incorporate other clinically important outcomes, such as morbidity, to improve relevance and applicability.
Acknowledgments
The authors would like to thank the medical and nursing staff of the Neonatal Intensive Care Unit (NICU) and the Clinical Pathology Laboratory of Dr. Kariadi General Hospital, Semarang, for their assistance in data collection and laboratory examinations. The authors also thank the Faculty of Medicine, Universitas Diponegoro, for the institutional support provided to this study.
Financial Disclosure
The authors received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors for this research.
Conflict of Interest
The authors declare no conflict of interest regarding the publication of this study.
Informed Consent
This study was based on a retrospective review of medical records and laboratory data. Because all data were analyzed anonymously and no direct contact with patients or families occurred, the requirement for written informed consent was waived by the institutional review board.
Author Contributions
Conceptualization: DRA and NSW; data collection: MSA; data analysis and data interpretation: JA; formal analysis: DRA and NSW; resources: MSA; data curation: JA; writing – original draft preparation: DRA and NSW; writing – review and editing: NSW; visualization: DRA; supervision: NSW; project administration: MRA; funding acquisition: JA. All authors have read and agreed to the published version of the manuscript.
Data Availability
The datasets generated and analyzed during the current study are not publicly available due to institutional policies on patient data confidentiality but are available from the corresponding author on reasonable request.
| References | ▴Top |
- World Health Organization. Guidelines on basic newborn resuscitation. WHO; 2012.
- World Health Organization. SDG target 3.2: End preventable deaths of newborns and children under 5 years of age [Internet [Internet]. 2022. Available from: https://www.who.int/data/gho/data/themes/topics/sdg-target-3-2-newborn-and-child-mortality.
- UNICEF. Indonesia country profile: Maternal and newborn health [Internet [Internet]. 2015. Available from: https://data.unicef.org/wp-content/uploads/country_profiles/Indonesia/country%20profile_IDN.pdf.
- Martono TU. Risk factors for birth asphyxia. Folia Medica Indonesiana. 2011;47(4):211-214.
- Albab MU, Widyaningsih H, Hartini S, Ambarwati A. Incidence of neonatal asphyxia events in mothers with premature ruptured amniotic fluid. South East Asia Nursing Research. 2020;2(3):99-104.
- Molace SM, Ahmed M, Kshirsagar VY, Bjapai R. Study of nucleated red blood cell counts in asphyxiated newborns and the fetal outcome. International Journal of Clinical Pediatrics. 2014;3(3):79-85.
- Morton S, Brettin K, Feldman H, Leeman K. Association of nucleated red blood cell count with mortality among neonatal intensive care unit patients. Pediatrics & Neonatology. 2020;61(6):592-599.
- Iribarren I, Hilario E, Alvarez A, Alonso-Alconada D. Neonatal multiple organ failure after perinatal asphyxia. Anales de Pediatria (English Edition. 2022;97(4):280.1-280.8.
- Ministry of Health of the Republic of Indonesia. National Clinical Practice Guidelines for the Management of Asphyxia. Jakarta: Ministry of Health of the Republic of Indonesia; 2019.
- Iqbal M, Razzaq A, Malik FA, Ali M, Imran M, Shahzadi M. Relationship of nucleated red blood cells with severity of perinatal asphyxia and its short-term outcome. Annals of Pakistan Institute of Medical Sciences. 2022;18(3):246-251
- Pikora K, Kretowska-Grunwald A, Krawczuk-Rybak M, Sawicka-Zukowska M. Diagnostic value and prognostic significance of nucleated red blood cells (NRBCs) in selected medical conditions. Cells. 2023;12(14).
doi pubmed - Krishnan KD, Avabratha KS, D’Souza AJ. Estimation of average birth weight in term newborns: A hospital-based study in coastal Karnataka. International Journal of Contemporary Pediatrics. 2017;1(3):156-159.
- Ardissino M, Morley AP, Slob EAW, Schuermans A, Rayes B, Raisi-Estabragh Z, de Marvao A, et al. Birth weight influences cardiac structure, function, and disease risk: evidence of a causal association. Eur Heart J. 2024;45(6):443-454.
doi pubmed - Li ZN, Wang SR, Wang P. Associations between low birth weight and perinatal asphyxia: A hospital-based study. Medicine. 2023;102(13):33137
- Mietzsch U, Kolnik SE, Wood TR, Natarajan N, Gonzalez FF, Glass H, Mayock DE, et al. Evolution of the Sarnat exam and association with 2-year outcomes in infants with moderate or severe hypoxic-ischaemic encephalopathy: a secondary analysis of the HEAL Trial. Arch Dis Child Fetal Neonatal Ed. 2024;109(3):308-316.
doi pubmed - Biedron N, Tokarzewska A, Rekowska A, Kwiatkowska A, Urbas W, Tarkowska A. Neonatal asphyxia - Predicting its occurrence and neurodevelopment of children after hypothermia treatment in the context of machine learning models: Literature review. Journal of Pre-Clinical and Clinical Research. 2024;18(1):26-32.
- Douvanas A, Kalafati M, Tamvaki E, Nieri A, Papalois A, Koulouglioti C, et al. Ventilation with or without endotracheal tube leak in prolonged neonatal asphyxia. Cureus. 2021;13(9):17798.
- Giannakis S, Ruhfus M, Markus M, Stein A, Hoehn T, Felderhoff-Mueser U, Sabir H. Mechanical ventilation, partial pressure of carbon dioxide, increased fraction of inspired oxygen and the increased risk for adverse short-term outcomes in cooled asphyxiated newborns. Children (Basel). 2021;8(6).
doi pubmed - Salvo V, Gazzolo D, Zimmermann LJ. The complex interrelationship between mechanical ventilation and therapeutic hypothermia in asphyxiated newborns. A review. Ther Hypothermia Temp Manag. 2024;14(2):80-88.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, including commercial use, provided the original work is properly cited.
International Journal of Clinical Pediatrics is published by Elmer Press Inc.